-
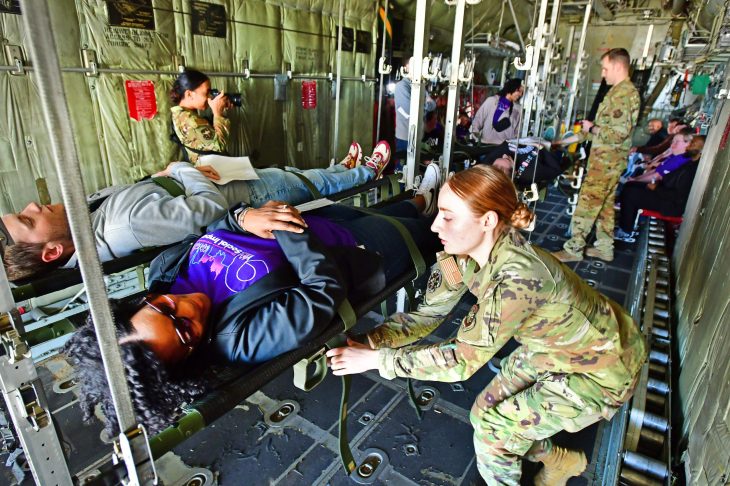 Ensuring Veteran care during crisis through lifesaving exercises
Ensuring Veteran care during crisis through lifesaving exercisesEach year, VHA’s Office of Emergency Management (OEM) conducts disaster-related exercises throughout the nation at strategic hubs, known as Federal Coordinating Centers (FCC), intended for receiving…
-
Ray & Mascari Inc. Recalls 4 Count Vine Ripe Tomatoes Because of Possible Health Risk
Ray & Mascari Inc. of Indianapolis, Indiana, is recalling 4 Count Vine Ripe Tomatoes packaged in clam shell containers [20 oz. (1 lb. 4 oz) 567g] with UPC# 7 96553 20062 1, and a master case label with…
-
In Reversal, FDA Rehires Staff Tasked With Releasing Public Records
The FDA has rehired at least some workers tasked with releasing public records generated by the agency’s regulatory activities, two employees said. The recall reverses…
-
Williams Farms Repack LLC Recalls Tomatoes Due to Possible Salmonella Contamination
Williams Farms Repack LLC is recalling Tomatoes sizes; 4×5 2 layer, 60ct 2layer, 3ct trays in the Williams Farms Repack label, and 5×6 25lb, 6×6 25lb H&C Farms Label, due to a potential contamination…
-
Patient Listening Session Summaries
Patient Listening Session summaries are published after each session to share a high-level summary of the discussion. The Public Engagement Staff draft summaries for FDA-requested sessions and the…
-
Requesting an FDA Patient-Led Listening Session
Learn about how to request a Patient Listening Session with the FDA to share your experiences and perspectives.
-
Suggested Sample Agenda for an FDA Patient-Led Listening Session
View a suggested sample agenda an FDA Patient-Led Listening Session
-
Planning an FDA Patient-Led Listening Session
Learn the steps for planning an FDA Patient-Led Listening Session.
-
Preparing Patient and Caregiver Participants for an FDA Patient-Led Listening Session and Suggested Topics
Learn about topics that are outside of the scope of FDA’s work and suggested topics for FDA-Patient-Led Listening Sessions.
-
Recommendations to Reduce the Risk of Transmission of Mycobacterium tuberculosis (Mtb) by Human Cells, Tissues, and Cellular and Tissue-Based Products (HCT/Ps)
This is the Draft Guidance for Industry – Recommendations to Reduce the Risk of Transmission of Mycobacterium tuberculosis (Mtb) by Human Cells, Tissues, and Cellular and Tissue-Based Products (HCT/Ps)
- Load More






