-
FDA Rare Disease Innovation Hub
FDA created the Rare Disease Innovation Hub (the Hub) to serve as a point of collaboration and connectivity between CBER and CDER with the goal of ultimately improving outcomes for patients.
-
I made a promise not just to myself, but to my community.
I pledged to be a Force for Health because I believe health isn’t just personal—it’s collective.I choose to LEARN what my body and mind need.
I choose to LIVE actively and make choices that fuel my future.
I choose to SHARE kindness and be a helping hand in my circle.
I choose to EARN…
-
appreciate it
-
Love it! Thanks for sharing!
-
-
-
dhirendralamsal2023gmail-com earned the togetherhood badge Getting Started 11 months ago
-
Turkana Food Inc. Recall Flora Dried Apricots with Undeclared Sulfites on Product Labeling Because of Possible Health Risk
Turkana Food Inc. Kenilworth, NJ is recalling 352 cases of Floria Dried Apricots because the product contains UNDECLARED SULFITES on the package label.
-
2025 Biological License Application Approvals
Listing of the 2025 Biological License Application Approvals
-
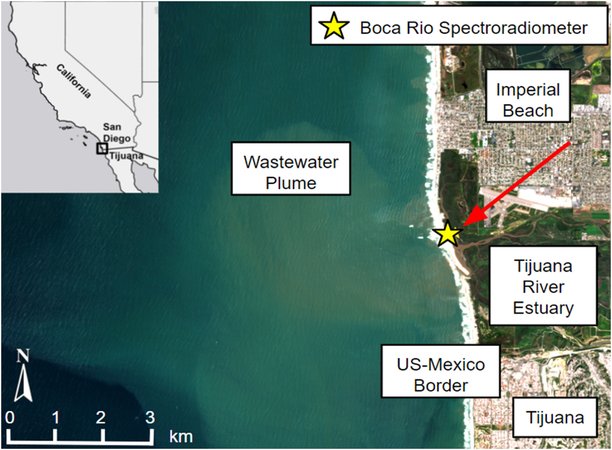 NASA Sensor on Space Station Eyes Contamination off California Coast
NASA Sensor on Space Station Eyes Contamination off California CoastAn instrument built at NASA’s Jet Propulsion Laboratory to map minerals on Earth is now revealing clues about water quality. A recent study found that…
-
Vita-Warehouse Corp. Issues Allergy Alert on Undeclared Peanut Allergen in ALDI Welby®, Berkley Jensen®, and VitaGlobe™ Vitamin B12 Gummy Products
June 12, 2025, Vita Warehouse Corp. is voluntarily recalling one lot of Welby® brand Vitamin B12 Energy Support gummy product 1000 mcg 140 gummies, Berkely Jensen® Vitamin B12 1000 mcg 250 Gummies, and…
-
Compliance Programs (CBER)
List of biological CBER compliance programs
-
- Load More








