-
Investigational New Drug Applications (INDs) for CBER-Regulated Products
An Investigational New Drug Application (IND) is a request for authorization from the Food and Drug Administration (FDA) to administer an investigational drug or biological product to humans.
-
Trump Turns Homelessness Response Away From Housing, Toward Forced Treatment
SACRAMENTO — President Donald Trump is vowing a new approach to getting homeless people off the streets by forcibly moving those living outside into large…
-
With Few Dentists and Fluoride Under Siege, Rural America Risks New Surge of Tooth Decay
In the wooded highlands of northern Arkansas, where small towns have few dentists, water officials who serve more than 20,000 people have for more than…
-
Consumer Updates
Science-based health and safety information you can trust.
-
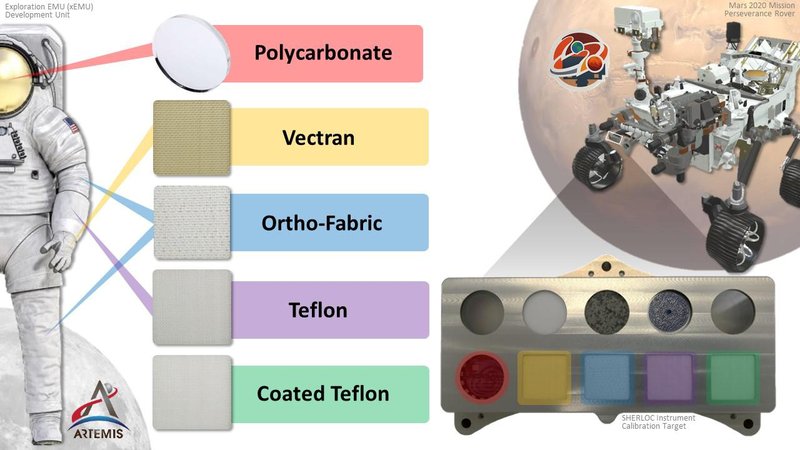 How NASA’s Perseverance Is Helping Prepare Astronauts for Mars
How NASA’s Perseverance Is Helping Prepare Astronauts for MarsNASA’s Perseverance rover landed on Mars in 2021 to search for signs of ancient microbial life and to help scientists understand the planet’s climate and…
-
Expanded Access to Experimental Biologics
Expanded access, sometimes called “compassionate use,” is the use outside of a clinical trial of an investigational medical product (i.e., one that has not been approved by FDA). FDA is committed to…
-
Approved Blood Products
Products that are regulated as Biologics License Applications (BLAs), New Drug Applications (NDAS), Premarket Approvals (PMAs), and 510(k)s.
-
Other Recommendations for Biologics Manufacturers
We have recently redesigned the FDA Web site. As a result, some Web links (URLs) embedded within Guidances, Rules and other documents are no longer valid. If you find a link that does not work, please…
-
Compliance Actions (Biologics)
CBER carries out a wide range of compliance and surveillance activities during the “life cycle” of biological products
-
Industry (Biologics)
CBER’s Manufacturers Assistance and Technical Training Branch provides assistance and training to industry, including large and small manufacturers and trade associations, and to respond to requests for…
- Load More






